Early QMSR Inspections: What FDA Is Finding
When the FDA’s Quality Management System Regulation (QMSR) formally replaced the legacy Quality System Regulation (QSR), medical device manufacturers knew that the regulatory framework would shift toward ISO 13485–aligned quality systems. What was less clear was how quickly FDA investigators would begin applying ISO concepts during inspections.
New inspection records from February 2026 provide an early glimpse into how QMSR is being implemented in practice. While the number of inspections is still small, the findings already reveal several important patterns about the types of compliance issues investigators are identifying.
A Different Kind of Inspection Record
One of the most notable differences in the early QMSR inspection data is how findings are documented.
Rather than a single narrative observation as typically seen in Form FDA 483 reports, the inspection records contain multiple entries tied to specific ISO 13485–aligned requirements. Each inspection may generate several clause-level findings, with separate records identifying the affected requirement and describing the deficiency.
This approach resembles an ISO audit structure, where each observation is mapped to a specific clause within the quality management system. It represents a significant shift from the historical QSR inspection model, which focused primarily on compliance with individual sections of 21 CFR Part 820.
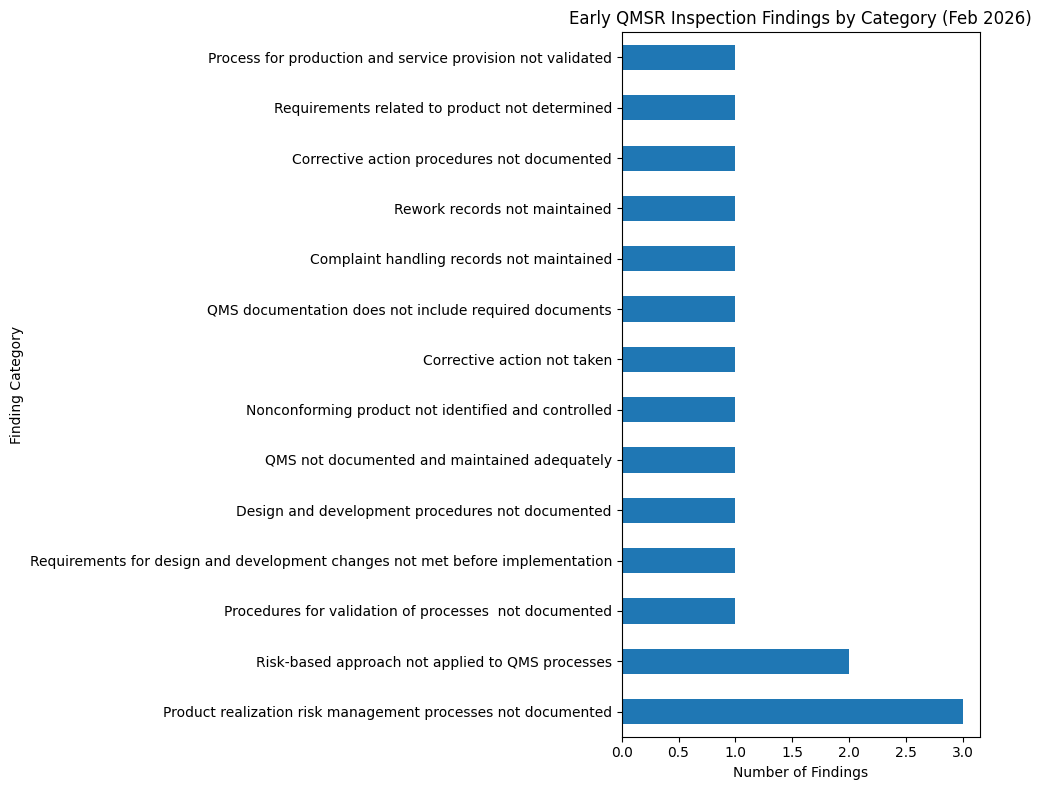
In the February dataset I reviewed, six inspections produced seventeen clause-level findings, averaging approximately 2.8 findings per inspection.
The Most Common Early Compliance Gaps
Although the sample size is small, several themes are already emerging from the findings.
Risk Management Integration
The most frequently cited issue involved failures to implement a risk-based approach to quality system processes. Several observations noted that manufacturers had not adequately incorporated risk management into their quality systems.
Under ISO 13485 and the QMSR framework, risk management is not confined to product design. Instead, it must be integrated throughout the quality system, including production controls, supplier management, and corrective action processes.
Early inspection findings suggest that some organizations have not yet fully adapted to this broader expectation.
Quality Management System Documentation
Another recurring issue involved incomplete or poorly defined QMS documentation. Investigators noted instances where required procedures or quality system elements were not adequately documented.
While documentation requirements are not new, ISO-based systems tend to emphasize process definition and system integration, which may expose weaknesses in legacy documentation structures originally designed for QSR compliance.
Design and Development Controls
Several findings related to design and development procedures, including inadequate documentation of design processes and insufficient control of design changes.
Design control deficiencies have long been a common issue in FDA inspections, and the early QMSR data suggest that this trend may continue under the new regulatory framework.
Corrective Action and Quality Oversight
Corrective action deficiencies also appeared in the findings. In some cases, investigators reported that corrective actions were not adequately implemented or that procedures governing corrective action were incomplete.
Corrective and preventive action systems remain a cornerstone of both QSR and ISO-based quality systems, and weaknesses in these processes often signal broader systemic issues.
Operational Recordkeeping
Additional findings involved operational recordkeeping, including incomplete complaint records and inadequate documentation of rework activities.
These issues reflect ongoing challenges with quality system discipline and documentation consistency, rather than fundamentally new regulatory expectations.
Early Signals from the Transition to QMSR
One important observation from these early inspections is that the deficiencies themselves are not dramatically different from historical QSR findings. Instead, the primary difference appears to be how the findings are framed.
Investigators are increasingly describing issues in terms of risk-based quality system processes, integrated system documentation, and defined process controls.
This language mirrors the structure and terminology of ISO 13485 audits, suggesting that FDA investigators are already approaching inspections from an ISO-aligned perspective.
In other words, the regulatory shift may be less about identifying entirely new types of violations and more about evaluating existing quality systems through a different conceptual framework.
What Manufacturers Should Watch
Although the early inspection dataset is limited, it highlights several areas where manufacturers should focus their transition efforts. The integration of risk management across the entire quality system, ensuring that QMS documentation is complete and coherent, strengthening design control procedures, maintaining robust corrective action processes, and improving operational documentation and recordkeeping.
These are foundational elements of both QSR and QMSR (ISO 13485) compliance. The difference under QMSR is that inspectors may now evaluate them through a more structured, system-level lens.
The Bottom Line
The first QMSR inspection records suggest that the transition to the new regulation may not immediately produce dramatically different enforcement patterns. Instead, investigators appear to be identifying familiar quality system weaknesses while framing them within an ISO 13485–aligned audit structure.
As more inspections occur under QMSR, these early signals will become clearer. For now, the initial findings reinforce a message that has long been true in medical device regulation. That a well-implemented, well-documented quality management system remains the strongest defense against regulatory risk.
Download the February 2026 Inspection Info
A Leap in FDA Transparency – The Electronic FOIA Reading Room
Exploring FDA’s FOIA Electronic Reading RoomA New Window into Inspection Findings The U.S. Food and Drug Administration (FDA) has expanded public access to regulatory information through its FOIA Electronic Reading Room, making large datasets from inspections and...
FDA Program to Accelerate U.S. Pharmaceutical Manufacturing
FDA Launches “PreCheck” Pilot Program to Accelerate U.S. Pharmaceutical Manufacturing The U.S. Food and Drug Administration has launched a new pilot initiative aimed at strengthening domestic pharmaceutical manufacturing and improving regulatory predictability for...
FDA Approves Novel Device for Pancreatic Cancer Treatment
FDA Approves Novel Device for Pancreatic Cancer Treatment The U.S. Food and Drug Administration has approved Optune Pax, a first-of-its-kind medical device designed to treat adult patients with locally advanced pancreatic cancer. Developed by Novocure, the portable...
When Apparel Becomes a Medical Device
Lessons from FDA’s Breast Binder Warning Letters In December 2025, the FDA issued warning letters to several companies selling breast binders and chest compression garments online. The agency concluded that marketing claims on company websites, like statements about...
FDA Issues 30 Warning Letters to Telehealth Firms
FDA Issues 30 Warning Letters to Telehealth Firms Over Compounded GLP-1 Marketing Claims On March 3, 2026, the U.S. Food and Drug Administration (FDA) announced that it has issued 30 warning letters to telehealth companies for making false or misleading claims...
FDA Warning Letter Cites Key CGMP and Data Integrity Failures
FDA Warning Letter Highlights Fundamental CGMP and Data Integrity Failures The FDA has issued a Warning Letter to A. Nelson & Co. Ltd. citing significant violations of Current Good Manufacturing Practice (CGMP) requirements under 21 CFR Parts 210 and 211 at a...